[17 Feb 2019]
Our manuscript Stimulus-driven brain rhythms within the alpha band: The attentional-modulation conundrum has just been accepted for publication in the Journal of Neuroscience. We show that stimulus-driven and intrinsic brain rhythms in the ~10 Hz range (alpha) can be functionally segregated. Briefly put, while one goes up the other one goes down.
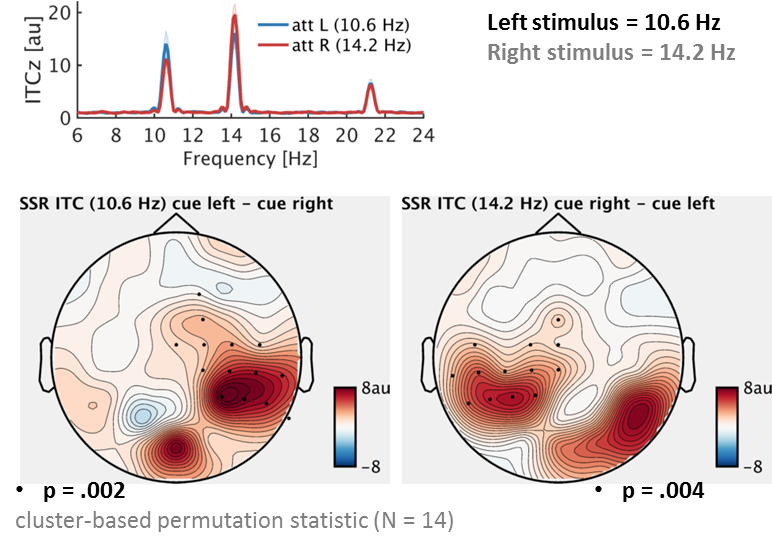
In an experiment, we recorded the brain waves of our participants while they were watching a screen with two stimuli. One, shown on the left, flickered at a rate of 10 Hz and another one, shown on the right, flickered at a rate of 12 Hz. (10 Hz flicker means that the stimulus cycles through a change in appearance or is simply switched on and off 10 times per second.) A very prominent notion has it that this type of visual stimulation is capable of taking possession, or “entrain”, the brain’s intrinsic alpha rhythm. The alpha rhythm can be characterised by its *amplitude* – the difference between peaks and troughs or, bluntly put, how strong it is – and its *phase* – when to expect a peak or trough based on its periodicity. From an entrainment perspective, alpha phase is assumed to lock on and align precisely to the periodicity of the visual stimulation.
Note that alpha itself has been looked into for almost a century and alpha phase has been tied to another exciting idea: How our brain processes visual input could be more akin to a camera than the continuous make-believe of our daily experience. Hereby, alpha works as a pacemaker that cuts the real-world continuity into perceptual samples or frames just like still frames of a movie. In line with this idea, experiments have shown that we seem to be less sensitive to “see” brief stimuli that pop up during one part of the alpha cycle – in the camera analogy, when the shutter is down – and more sensitive during another part, i.e. when the shutter is open.
Now, the *combination* of perceptual sampling and entrainment puts experimenters in a formidable position to study alpha’s role in perception. It allows them to manipulate alpha phase and exactly time the presentation of stimuli accordingly. Being able to entrain alpha (or other rhythms) through rhythmic visual stimulation would thus be a versatile and easy-to-apply tool – but does it really work?
In short, our experiment adds to a line of recent studies that challenge a straightforward alpha entrainment using visual flicker. Our main assumption was this: If it looks like alpha and behaves like alpha, then it should be alpha. *It* refers to the brain waves elicited by watching a 10 Hz flicker. Because the brain response shows up as 10-Hz rhythm in the EEG it does *look* like alpha – especially if you look at it in the frequency domain where it produces a neat 10-Hz spectral peak. “Does it behave like alpha?” we translated into “Does it have the same function?”
One very well documented effect is that alpha power (its strength) shifts according to where we attend to. If we focus our attention somewhere to our left (without actually looking there) then alpha power will go down in our right visual brain – due to the cross-wiring of our visual system from eye to cortex, this is where our left visual world is processed. This alpha decrease works like opening the gates for visual input to venture into further stages of processing. Simultaneously, alpha *increases* in the left visual brain, figuratively closing the gates to unattended, irrelevant sights to our right.
Would a brain response driven by our 10/12 Hz stimulation show a similar effect? If so, that would be strong evidence for a close relationship of spontaneous and stimulus-driven alpha brain waves. Using rhythmic flicker to control alpha experimentally would seem like a readily available manipulation. That was not what we found though. On the contrary – we were able to switch between alpha and the stimulus-driven brain waves using slightly different data analysis approaches. Also, attention had the known suppressive effect on alpha while the corresponding (i.e. same-side) stimulus-driven brain response increased.
These results led us to conclude that we are looking at two concurrent neural phenomena, alpha and flicker-driven brain responses. And each one of them seems to provide us with a different perspective on how attention alters our perception.
Find the specifics and references here.
Note 1: Of course, our results do not rule out alpha entrainment, only that an alpha range stimulus-driven brain wave should not be regarded as sufficient to show alpha entrainment. In the paper (and previous literature) we discuss several, possibly additional conditions that need to be satisfied to give rise to the phenomenon.
Note 2: Data and code to reproduce our results are available here. With minor modifications this code should be applicable to other datasets.
Disclaimer: Views expressed in this digest are mine (CK) and not necessarily shared in all their nuances between the co-authors of the manuscript.